IN Brief:
- Brady EMEA and Caretag are rolling out an integrated surgical instrument management offer across Europe.
- The system combines identification, RFID-based tray verification, and lifecycle traceability from sterilisation through theatre use and return.
- The partnership is positioned around efficiency gains, inventory visibility, and MDR-aligned documentation.
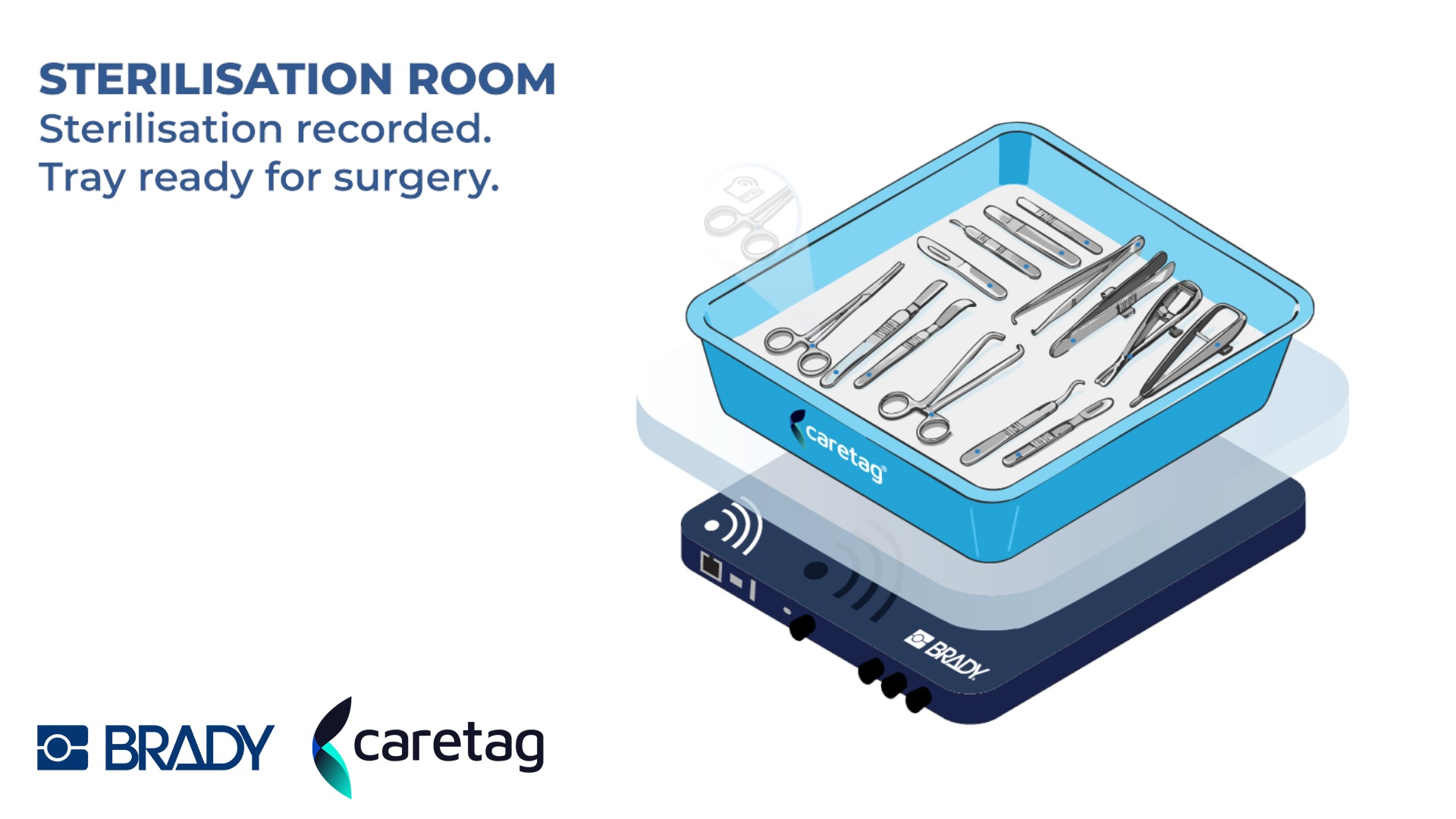
Brady EMEA and Caretag have entered a strategic partnership to deliver end-to-end surgical instrument tracking for hospitals and sterile supply departments across Europe, combining identification and data-capture technology with UHF RFID-based instrument management. The move brings two established parts of the healthcare workflow closer together at a time when providers are under growing pressure to document instrument handling more precisely and reduce waste across expensive surgical inventories.
The combined offer is designed to track instruments from sterilisation through operating-theatre use and back again, with full digital traceability across the cycle. Caretag said its RFID-enabled system allows simultaneous identification of multiple instruments and can accelerate instrument packing by more than 33% compared with conventional DataMatrix scanning. The company also said its tags are built to withstand more than 2,500 sterilisation cycles, while Brady contributes identification, labelling, printing, lasering, engraving, and data-capture capability for demanding clinical environments.
That combination is aimed at a familiar problem in hospital logistics. Surgical instrument management is still vulnerable to manual workarounds, partial records, and delays in locating or verifying trays, especially where high turnover and compliance pressure sit alongside limited staffing. Instrument sets carry both cost and clinical risk. If the chain of identification breaks down, the result can be missing items, slower turnaround, duplicated procurement, or uncertainty over whether a tray is complete and ready for use.
The timing also reflects the regulatory backdrop. EU MDR requirements and associated UDI obligations continue to tighten expectations around device traceability and documentation, with transition milestones extending through 2027 and 2028 depending on device class. In that environment, traceability systems are no longer peripheral IT purchases. They are moving toward the operational core of sterile processing, theatre readiness, and audit preparation.
What makes the Brady-Caretag arrangement notable is that it treats healthcare logistics as a full lifecycle problem rather than a point solution. Hospitals are increasingly looking for systems that can link identification, location awareness, usage history, and inventory planning without forcing staff into more manual work. The same data that proves compliance can also reveal underused assets, repeated shortages, and purchasing patterns that do not match real demand.
That is where these systems begin to move beyond simple traceability. Once instruments can be identified in bulk, tracked in motion, and tied back to documented workflows, the data starts to shape stock policy, maintenance planning, and procurement decisions. For providers trying to improve resilience without carrying unnecessary inventory, that is likely to become the more important part of the story.